
NZ Landcare Trust coordinator Alastair Cole examines a koura (freshwater crayfish) with a cit sci workshop participant
The final working group meeting for the Manawatu/Wairarapa took us to Pukaha/Mt Bruce, a 942ha remnant of lowland podocarp forest, 27km north of Masterton. The location was ideal for running a course on stream monitoring methods: nearly two decades of restoration have resulted in native dominated canopy and healthy waterways with diverse aquatic fauna. The education/visitors centre provided shelter and importantly, hot food and drinks on a wintry day.
As with previous events in this 3-yr New Zealand Landcare Trust-led project, participants were extremely diverse. Members from community groups and Environment Network Manawatu attended along with children relishing the opportunity to get wet. Fish & Game, Agresearch and Horizons Regional Council staff lead presentations and practical demonstrations – waders were the clothing du jour.
Stream monitoring methods
Three main methods were covered: Trapping, electrofishing and spotlighting. Each provides different types of data; skill levels vary as do the type and cost of equipment. In addition, not all methods suit all species of fish or stream conditions, so data may be biased (see table). Overall, there are synergies with the Biodiversity Monitoring Guide for Auckland Community groups (see posts here and here), given the need for groups to identify which method best suits their restoration objectives and the level of resourcing they have available.

Freshwater fish sampling protocols
The comprehensive and easy to read New Zealand Freshwater Fish Sampling Protocols covers the following three methods in detail. For more information on native fish, check out NZ Landcare Trust’s Fish Fact Sheet series (in English and te reo).
Electrofishing
Electrofishing is undoubtedly a specialist activity: you need to attend a training course e.g., via NIWA ($800 – $1000), and the equipment is expensive (c.15k). The purpose of electrofishing is to determine exactly what occurs in a given stream reach, allowing a finer grained analysis than e.g., spotlighting would provide. Using this method depends on the kind of data required, and the level of detail needed to answer the research/restoration question. An easier (and cheaper) solution is to approach Regional Councils to see if electrofishing can be done on behalf of the group.
Spotlighting
Spotlighting is useful for detecting many of our larger whitebait species (galaxiids) e.g., giant kokupu because they are mostly nocturnal. The method is fairly quick and non-invasive but becomes time consuming if fish need to be caught to accurately measure their sizes.
Trapping
Fyke nets and Gee minnow traps set out the day before yielded several long fin eels as well as the odd bully and juvenile trout. One challenge with this method that eels will eat other fish caught, but nets are available with exclusion pipes to avoid ending up with a catch of well-fed eels and (nothing else!). A wide range of traps designs are available, so being consistent with trap type will support the collection of comparable data.
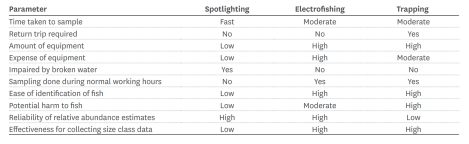
Guide to the relative advantages and disadvantages of protocol methods. From the NZ Freshwater Fish Sampling Protocols (Part 1) Joy, David and Lake (2013)
Community stream health assessment
The Stream Health Monitoring and Assessment Kit (SHMAK) and the Auckland Waicare programme are two examples of resources available to the community. SHMAK was developed by NIWA scientists in 2002 and is now being revitalised through a citizen science programme led by Dr. Richard Storey. Waicare, up until recently, had a team of dedicated coordinators actively worked with community members and school children to collect data on their local waterways and provide educational experiences (Waicare is now moving to a community-led model). Both SHMAK and Waicare cover macroinvertebrate identification; tools for measuring e.g., water clarity, conductivity, pH, temperature and flow; guides for assessing stream characteristics (e.g., substrate type) as well as habitat characteristics (e.g., riparian condition). All put together, the measures provide a snapshot of stream and surrounding habitat health.

Sampling stream invertebrates at Mt Bruce/Pukaha
Measuring E. coli
Dr Adrian Cookson (AgResearch) outlined current research looking at determining the difference between environmental and animal gut derived E. coli. This has implications for how areas are managed, as it will help pin point sources. This is highly specialised work, but for community members, two basic methods for detecting E. coli in general look promising.
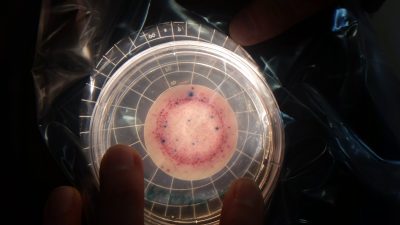
E. coli sample: colonies are dyed blue
Friends of the Matai (Nelson) are currently comparing Petrifilm plates (supplied by NIWA) with the Sanita-kun kits (supplied by Ngaio Diagnostics). The third kit, Aquagenx, was discarded early on because readings were at the top of its range, and were difficult to use (and very smelly!).
The results from petrifilms and Sanita-kun are comparable. All methods use membrane filtration and then placing the filter paper on a growth medium, incubating for 24 hours and counting the colonies. The petrifilm method uses 100ml individual single use filter pots. The water sample is drawn through the filter using a syringe. The Sanita-kun method uses glassware which can be washed and reused.
The setup cost for Sanita-kun is greater owing to the glassware but ends up c. half the price of the petrifilm. Although further results are still to be analysed, Philippa Eberlein (FOM Member) highlights the most practical option appears to be Sanita-kun glassware combined with the petrifilm plates.
Databases…
The NZ Freshwater Fish Database is housed at the National Institute of Water and Atmosphere (NIWA). Registered users download field sheets and then re-enter their data onto the website when convenient. Entering data directly relies on web coverage, so the development of an app would be a better option. Although all entries are checked by experts, manual translation from field sheets to online database is time consuming and raises the potential for error (e.g., misinterpreted notes), field sheets being lost, made illegible, or simply not inputted. There is great potential here for citizen science… examples include anglers in Ireland logging their catches and a global freshwater fish BioBlitz.
Native vs exotic…
With all workshops, there is plenty of time for discussion. Brown trout were introduced 150 years ago (!), and are present at Pukaha. Fish and Game rep Matt Kavermann highlighted the lack of known extinctions of native species known occurring from brown trout. Yes, they have caused population declines as well as restrictions in ranges of native species, but at the same time trout fishing overall contributes more than $100 million/yr – that’s to the Taupo economy alone. At Pukaha, the emphasis is on managing the trout population, rather than eradication.

My brother and I with our rainbow trout catch at Lake Rotorua, 1980. Nowadays, I’d describe this activity as a form of recreational predator control!
With public interest and concern for our freshwater resources increasing, learning basic monitoring skills helps raise awareness of stream health state and trends and has the potential to build more complete data sets of our freshwater resources. Our small workshop is one of many underway around NZ. We still have three more working group meetings to go (in Auckland, Nelson and Dunedin), and each will have a practical citizen science training emphasis. The 3-yr project will wrap up with a one-day citizen science symposium in Wellington… more about that in later posts!

Pingback: Final Dunedin #citizenscience Working Group Meeting: Urban #freshwater #monitoring | monicalogues·
Pingback: #citizenscience in Nelson: Stream monitoring for community groups, schools and farmers | monicalogues·